Issue 3: New Insights into Muscle Health
A muscle fiber fails, stops performing as it once did.
No rupture. No trauma. No headline injury–>Force production weakens, neuromuscular signaling frays, and metabolic efficiency slips. The fiber becomes less useful, more costly to maintain.
At this point, the immune system has an important decision to make:
Is this tissue worth repairing?
Or is it better cleared, recycled, and replaced elsewhere?
The Immune Intelligence of Muscle Loss
Sarcopenia begins not when muscle disappears, but when muscle is quietly deprioritized.
1. Muscle Loss Is an Immune Act, Not a Metabolic Accident
We often describe muscle loss in accounting terms:
breakdown exceeds synthesis, catabolism outpaces anabolism.
But this language hides the central truth.
Muscle loss is not passive. It is authorized.
The immune system plays a decisive role in determining whether muscle tissue is:
- repaired
- remodeled
- tolerated in a diminished state
- or actively dismantled
This is true in acute injury, chronic disease, aging, and cachexia — the difference is not whether immunity is involved, but how strongly and how persistently it signals.
Why muscle is subject to immune judgment
Muscle is one of the most metabolically expensive tissues in the body. It demands:
- continuous energy supply
- intact innervation
- structural maintenance
- coordinated repair
From an evolutionary standpoint, this makes muscle a conditional asset.
In states of:
- infection
- chronic inflammation
- malignancy
- systemic stress
- aging with reduced reserve
the immune system may interpret muscle preservation as nonessential, or even maladaptive. This is not dysfunction. It is triage.
Sarcopenia vs. cachexia: same logic, different intensity
Sarcopenia and cachexia are often discussed as distinct entities. Immunologically, they sit on the same continuum.
- Sarcopenia reflects:
- low-grade, chronic immune activation
- incomplete repair signaling
- prolonged “maintenance mode” without regeneration
- Cachexia reflects:
- high-grade systemic inflammation
- overwhelming catabolic signals
- immune-driven dismantling of muscle and fat
In both cases, muscle loss is not a failure of effort or nutrition alone.
It is the outcome of an immune system that has shifted its priorities.
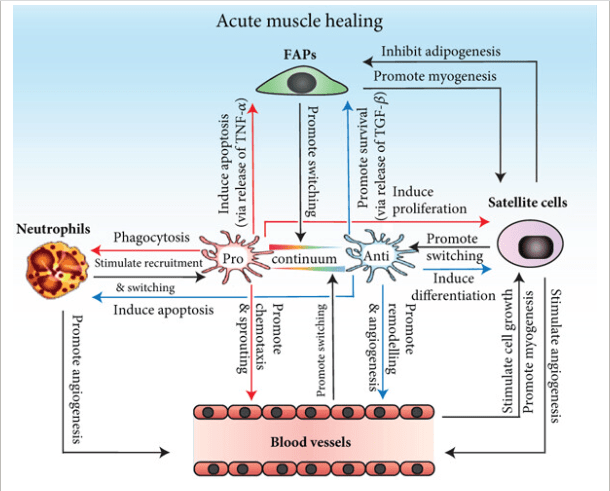
2. Macrophage Polarization: Where Repair Is Chosen—or Denied
If muscle loss is an immune act, macrophages are the decision-makers.
Macrophages are not simply scavengers. They are interpreters of context — cells that read the environment and decide what comes next.
The normal repair sequence
After muscle stress or injury, a healthy repair cycle unfolds in stages:
- M1 macrophages arrive first
- pro-inflammatory
- debris-clearing
- secrete cytokines such as TNF-α and IL-1β
- essential for removing damaged tissue
- M1 → M2 transition
- inflammation resolves
- signaling shifts
- M2 macrophages dominate
- pro-repair
- support satellite cell activation
- promote myogenesis, angiogenesis, and ECM remodeling
This transition is not optional.
Without it, muscle cannot regenerate.
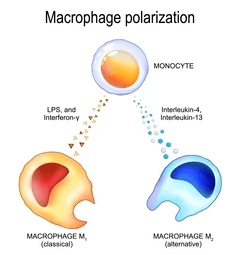
“M1” and “M2” Macrophages: A Useful Fiction
The terms M1 and M2 macrophages are widely used in the scientific and medical literature — but they are shorthand, not biological absolutes.
There are no discrete “M1 cells” and “M2 cells” in the body.
Instead, macrophages exist along a dynamic functional spectrum, adapting their behavior in response to local signals, tissue context, and systemic state.
What the terms are trying to capture
- “M1-like” macrophages
- prioritize pathogen defense and debris clearance
- secrete pro-inflammatory cytokines (e.g., TNF-α, IL-1β)
- promote catabolic and stress-response programs
- are essential in the early phase of injury or stress
- “M2-like” macrophages
- support tissue repair and regeneration
- promote angiogenesis, ECM remodeling, and stem-cell activation
- secrete anti-inflammatory and pro-resolution signals
- dominate during the recovery phase
These labels describe dominant functional tendencies, not fixed identities.
Why the distinction still matters
Despite its limitations, the M1/M2 framework remains useful because it highlights a critical biological truth: Successful tissue repair depends not on inflammation alone, but on the timely resolution of inflammation.
In muscle, regeneration requires:
- an initial inflammatory response
- followed by a phenotypic shift toward repair-supportive macrophage behavior
Failure of this shift — not absence of immune cells — is a major driver of sarcopenia and impaired recovery.
What goes wrong in chronic disease and aging
In aging, chronic illness, and persistent stress states:
- macrophages remain biased toward inflammatory signaling
- resolution cues are blunted or ignored
- repair programs are delayed or never fully engaged
Muscle is not aggressively destroyed — it is simply never given permission to rebuild.
Why Wellth-e uses the terms anyway
M1 and M2 are imperfect language for a complex reality.
But they provide a shared conceptual handle for understanding how immune tone — not just immune presence — determines whether muscle is preserved, repaired, or slowly lost.
Macrophages don’t switch identities.
They switch priorities.
*For an overview of macrophages, you can watch my presentation at IHMC for a lay audience: https://youtu.be/-CQqfkrae00?si=Lkuqlyi18NamHaLg
What goes wrong in sarcopenia
In aging and chronic disease, the problem is rarely the absence of macrophages. It is the failure to change phenotype.
Several forces stall the M1 → M2 transition:
- chronic low-grade inflammation (“inflammaging”)
- persistent sympathetic activation
- metabolic acidosis
- hormonal deficiency
- poor sleep and recovery
- repeated stress without resolution
The result is a muscle environment stuck in perpetual cleanup mode.
Debris is cleared.
But rebuilding never begins.
Why stalled repair leads to net muscle loss
Prolonged M1 dominance creates a paradoxical state:
- inflammation is not severe enough to trigger acute repair
- but persistent enough to block regeneration
Satellite cells receive mixed signals.
ECM remodeling becomes maladaptive.
Protein synthesis is blunted — not because nutrients are absent, but because repair is not authorized.
Over time, the immune system quietly concludes: This tissue is no longer worth the investment.
And muscle mass and function decline slowly, steadily, and often invisibly.
A crucial reframing
This is why sarcopenia so often resists:
- more protein
- more supplements
- more training
Those inputs matter — but only after the immune environment allows repair.
Muscle does not grow in a state of unresolved immune surveillance.
Phagocyte Takeaway
Muscle loss is not simply something that happens to muscle.
It is something the immune system permits.
Until we understand sarcopenia as a failure of immune resolution — not just a deficit of calories, hormones, or effort — we will continue to treat the symptom while missing the decision.
3. The Immune–Stem Cell Conversation: When Repair Is Possible—and When It Isn’t
One of the most persistent myths about sarcopenia is that aging muscle “runs out” of stem cells.
It doesn’t.
In most cases, satellite cells are still present — sometimes in surprisingly normal numbers. What fails is not their existence, but the conversation that tells them what to do.
Muscle regeneration is not a solo act. It is a dialogue among:
- immune cells
- stem cells
- the extracellular matrix (ECM)
- and mechanical signals from loading and movement
When that dialogue is coherent, repair proceeds. When it’s distorted, regeneration stalls.
The Satellite Cell: Poised but Conditional
Satellite cells reside in a very specific anatomical niche:
between the muscle fiber membrane (sarcolemma) and the basal lamina, a specialized layer of the ECM.
This location is not incidental. It places satellite cells at the intersection of:
- mechanical strain from contraction
- immune-derived signals
- matrix-bound growth factors
In healthy muscle, satellite cells cycle through three tightly regulated states:
- quiescence (protected, metabolically quiet)
- activation and proliferation (in response to injury or load)
- differentiation and fusion (repairing or adding to muscle fibers)
The immune system plays a decisive role in determining whether this cycle advances or arrests.
Macrophages as Instructors, Not Just Cleaners
During successful repair, macrophages do more than clear debris.
Repair-supportive (M2-like) macrophages:
- secrete growth factors such as IGF-1
- release cytokines that promote myogenesis
- modulate ECM remodeling
- provide timing cues that allow satellite cells to differentiate
These signals do not simply say “grow.”
They say “now is the right moment to grow.”
When macrophage signaling remains inflammatory or unresolved, satellite cells receive mixed instructions:
- proliferate but do not differentiate
- remain quiescent despite injury
- or divert toward non-myogenic fates
This is how muscle becomes populated with stem cells that are present but ineffective.
The ECM: Not a Scaffold, but a Script
The extracellular matrix is often described as structural support. In reality, it is a signaling environment.
The basal lamina:
- binds growth factors
- transmits mechanical strain
- regulates stiffness and elasticity
- shapes how immune and stem cells interpret local conditions
When the ECM is healthy, it:
- maintains satellite cell quiescence until needed
- releases bound signals during mechanical loading
- supports orderly regeneration
With aging, chronic inflammation, and inactivity:
- collagen cross-linking increases
- fibrosis accumulates
- matrix stiffness rises
- signaling gradients become distorted
Satellite cells still “hear” signals — but they are misleading. Repair becomes inefficient, incomplete, or misdirected.
From Failed Repair to Sarcopenia
When immune resolution is impaired and ECM signaling is distorted, muscle enters a chronic liminal state:
- not acutely injured
- not actively regenerating
- slowly losing functional fibers
This state favors:
- replacement with fibrotic tissue
- infiltration of fat
- reduced force transmission
- impaired neuromuscular signaling
Over time, the immune system adapts to this new baseline.
Muscle loss is no longer treated as damage. Rather, it’s seen as acceptable attrition by the body.
Why PRP and “Stem Cell” Therapies Often Fail
Regenerative therapies such as platelet-rich plasma (PRP) and so-called “stem cell injections” are frequently framed as ways to add repair capacity to damaged tissue.
But repair does not fail in sarcopenia because stem cells are absent.
It fails because the environment is hostile to regeneration.
The core problem: context, not content
In aging muscle, chronic disease, or cachexia:
- inflammation is unresolved
- macrophage signaling remains biased toward catabolism
- the extracellular matrix is fibrotic or overly stiff
- satellite-cell instructions are distorted
Injecting growth factors or cells into this environment does not override those signals.
It places regenerative elements back into a system that has already decided not to rebuild.
Why PRP may disappoint
PRP delivers a burst of growth factors.
But:
- growth factors require a receptive immune and ECM context
- without immune resolution, signals are transient
- repeated PRP does not reprogram macrophage behavior
In chronically inflamed tissue, PRP may produce short-lived symptom relief without durable structural repair.
Why many “stem cell” treatments underperform
Most commercially available “stem cell” procedures do not implant true muscle stem cells capable of long-term engraftment.
Even when viable progenitors are present:
- they depend on ECM cues to survive and differentiate
- inflammatory cytokines can suppress or redirect their fate
- stiff, fibrotic matrix discourages myogenesis
The cells themselves are not defective. The instructions they receive are.
A systems-level truth
Regeneration is not additive.
You cannot simply add:
- cells
- growth factors
- biologics
to a system that is locked in a catabolic immune state and expect durable repair. First, the immune system must signal: This tissue is worth saving. Only then do regenerative therapies have a chance to succeed.
Why this matters for sarcopenia
This explains why:
- regenerative injections show variable results
- outcomes are worse in older or chronically ill patients
- mechanical loading, sleep, and recovery often matter more than biologics
True regeneration requires:
- immune resolution
- ECM remodeling
- mechanical signaling
- metabolic support
Without these, “regenerative” therapies are often temporizing at best.
A Crucial Reframe
Sarcopenia is often described as diminished anabolic response.
At a deeper level, it is a failure of permission.
The immune–stem cell–matrix axis no longer agrees that muscle repair is worth the investment.
Until that agreement is restored — through resolution of inflammation, mechanical signaling, and appropriate recovery — nutrients and exercise alone will struggle to reverse the process.
Takeaway
Muscle stem cells do not disappear with age. They become misguided.
Repair fails not because the machinery is gone, but because the instructions are corrupted.
Understanding sarcopenia means understanding who writes those instructions — and when they are allowed to change.
4. Atrogenes: How the Immune System Authorizes Dismantling
Muscle loss is not merely the absence of growth.
It is the presence of an active dismantling program.
That program is executed through a set of genes collectively known as atrogenes — molecular instructions that tell muscle fibers how to disassemble themselves efficiently and irreversibly.
Two names appear repeatedly in this process:
- Atrogin-1 (MAFbx)
- MuRF-1 (Muscle RING Finger-1)
These are not markers of damage but effectors of intent.
What atrogenes actually do
Atrogin-1 and MuRF-1 encode E3 ubiquitin ligases — enzymes that tag specific muscle proteins for degradation by the proteasome.
They preferentially target:
- myosin heavy chains
- sarcomeric structural proteins
- regulators of muscle growth and differentiation
When these genes are activated:
- contractile machinery is dismantled
- force production declines
- fibers shrink and weaken
This is not random decay. Your body executes this program because it is selective, energy-efficient recycling.
What turns atrogenes on
Atrogene activation is tightly regulated, and immune signaling is central to that regulation.
Key activators include:
- TNF-α
- IL-1β
- chronic IL-6 signaling
- glucocorticoids
- metabolic acidosis
- denervation or impaired NMJ signaling
These signals converge on transcriptional regulators such as:
- FOXO
- NF-κB
Once engaged, these pathways shift muscle into a catabolic state, even in the presence of adequate nutrition.
This is why sarcopenia so often persists despite protein intake: the degradation machinery is already running.
Atrogenes as immune logic, not failure
It is tempting to view atrogene activation as pathology.
But from an immune perspective, it is logic.
In conditions of:
- chronic inflammation
- systemic illness
- aging with limited reserve
muscle becomes an expendable store of amino acids.
Atrogenes allow the body to:
- reclaim nitrogen
- support immune function
- fuel gluconeogenesis
- reduce metabolic burden
This is not the body “breaking down.” but intelligently reallocating resources.
Why sarcopenia is slow — and cachexia is fast
The difference between sarcopenia and cachexia is not which genes are used, but how loudly and how long they are activated.

- Sarcopenia
- low-grade, persistent atrogene activation
- incomplete shutdown of catabolic programs
- gradual erosion of muscle mass and power
- Cachexia
- high-grade systemic inflammation
- sustained, overwhelming atrogene signaling
- rapid loss of muscle and fat
- loss becomes life-threatening
Both are immune-authorized. It’s just a matter of intensity.
Why this reframes treatment
If muscle loss is driven by active atrogene programs, then:
- protein alone cannot stop it
- supplements alone cannot override it
- exercise without immune resolution may fail
Effective reversal requires turning off the dismantling signal.
That means addressing:
- inflammatory tone
- hormonal suppression
- denervation and NMJ integrity
- sleep and recovery
- mechanical loading that signals usefulness
Until the immune system decides muscle is worth preserving, atrogenes remain engaged.
The hidden cost of ignoring this
When sarcopenia is treated as passive wasting:
- clinicians chase nutrition
- patients blame effort
- systems miss the signal
But when it is recognized as an active immune program, the strategy changes.
The question becomes:What conditions would persuade the immune system to stop dismantling this tissue?
That is the real therapeutic target.
Takeaway
Muscle loss is not a failure of construction but the execution of a dismantling order (demolition).
Atrogenes activate when the immune system concludes that muscle is no longer worth the cost.
Understanding sarcopenia requires asking not how muscle is lost, but why the body decides to let it go.
5. Signals Without Borders: How Muscle Is Reprogrammed From Afar
One of the most unsettling features of sarcopenia and cachexia is that muscle can waste without being injured, unloaded, or directly inflamed.
A patient with cancer loses muscle in limbs never touched by chemotherapy.
A person with heart failure loses muscle far from the failing myocardium.
An older adult loses strength despite “doing everything right.”
This is not local failure but remote instruction. Muscle is being told — at a distance — to stand down.
Endocrine Immunity: When Inflammation Travels
The immune system does not operate solely at the site of damage. It communicates system-wide through circulating signals that carry context, not just content.
Key among these are:
- cytokines
- hormones
- metabolites
- and increasingly recognized: extracellular vesicles (EVs)
These signals do not merely report inflammation but actively rewrite priorities in distant tissues.
Extracellular Vesicles: Immune Messages in Motion
Extracellular vesicles are membrane-bound packages released by cells, including:
- adipocytes
- immune cells
- tumor cells
- stressed tissues
They carry:
- microRNAs
- proteins
- lipids
- transcriptional regulators
Unlike free-floating cytokines, EVs are:
- protected from degradation
- selectively targeted
- capable of altering gene expression in recipient cells
They are instructions.
Adipose Tissue as a Signaling Organ
Adipose tissue — particularly inflamed adipose tissue — is not inert energy storage. It is an active immune organ.
Adipose tissue macrophages (ATMs) release EVs that have been shown to:
- alter insulin sensitivity
- reprogram mitochondrial function
- modify epigenetic marks in distant tissues
In models of obesity and chronic disease, ATM-derived EVs:
- promote catabolic signaling in skeletal muscle
- impair oxidative metabolism
- favor atrogene activation
Muscle receives the message that energy is scarce and is instructed to preserve the system, not the tissue.
Cancer Cachexia: The Extreme Case
In cancer, this process is amplified.
Tumors release EVs that:
- suppress protein synthesis
- activate FOXO pathways
- increase ubiquitin–proteasome activity
- impair mitochondrial function
These effects occur even when:
- nutrition is adequate
- muscle is unloaded
- local inflammation is absent
Cachexia is not “starvation”. It is systemic reprogramming driven by immune and tumor-derived signals that treat muscle as expendable.
Why Muscle Cannot “Opt Out”
Muscle does not decide its own fate.
It responds to:
- immune tone
- hormonal environment
- systemic stress signals
When EV-mediated messaging and cytokine signaling converge, muscle fibers:
- reduce anabolic sensitivity
- activate degradation pathways
- suppress repair programs
Even aggressive resistance training can be overridden if the systemic signal is strong enough.
This explains why:
- cachexia is resistant to nutrition alone
- sarcopenia progresses despite effort
- regenerative therapies fail without immune recalibration
The Immune Logic Behind Remote Wasting
From the immune system’s perspective, this makes sense.
In chronic disease:
- immune surveillance is prioritized
- glucose and amino acids are redirected
- tissues with lower immediate survival value are downregulated
Muscle is not being punished but reassigned.
Why This Matters Clinically
Once we recognize that muscle can be instructed to waste remotely:
- sarcopenia can no longer be treated as a local problem
- cachexia can no longer be framed as nutritional failure
- rehabilitation must address systemic immune signals
This also explains why:
- anti-inflammatory strategies sometimes restore anabolic response
- treating the underlying disease can reverse wasting
- muscle mass rebounds only after immune tone shifts
Takeaway
Muscle loss does not require injury; it requires instruction.
That instruction can be granted from afar — by immune cells, adipose tissue, tumors, or chronically inflamed systems that decide muscle is no longer worth the cost.
Sarcopenia and cachexia are not isolated failures of muscle. They are system-wide decisions, delivered quietly, efficiently, and at a distance.
6. Reversing the Signal: What Actually Changes Immune Decisions
If muscle loss is an immune-authorized process, then reversal cannot rely on force alone. You cannot argue muscle back into existence. You must change the signals that led the immune system to deprioritize it.
This is where many interventions fail, not because they are ineffective, but because they are aimed at the wrong level of the system.
The Core Principle
The immune system does not ask: How hard are you training?
It asks: Is this tissue worth protecting under current conditions?
To reverse sarcopenia or blunt cachexia, the body must be convinced of three things:
- The stress is survivable
- The tissue is useful
- Repair is affordable
Only when these criteria are met does immune tone shift from dismantling to preservation.
What Actually Moves the Needle
1. Resolution of Inflammation — Not Suppression
Blunting inflammation indiscriminately can impair repair.
What matters is resolution:
- timely M1 → M2 macrophage transition
- clearance of debris without persistent signaling
- restoration of immune flexibility
This is why:
- chronic NSAID use can impair muscle adaptation
- immune-calming strategies outperform immune-silencing ones
- sleep and circadian alignment matter as much as training
2. Mechanical Use That Signals Value
Muscle is judged by utility, not intention.
Mechanical signals that persuade the immune system include:
- tension without excessive damage
- repeated, predictable loading
- neuromuscular engagement with coordination and intent
This is why:
- low-load, high-tension work can preserve muscle in frailty
- isometrics and slow eccentrics are disproportionately effective
- erratic, punishing training can worsen immune resistance
The message must be: This tissue is being used–and used well.
3. Neuromuscular Integrity
The immune system is exquisitely sensitive to denervation.
Impaired NMJ signaling:
- increases inflammatory tone
- activates atrogenes
- biases macrophages toward catabolism
Restoring neuromuscular signaling — through movement quality, coordination, and recovery — changes immune interpretation.
Muscle that communicates clearly with the nervous system is protected.
4. Energetic Sufficiency Without Excess Alarm
Muscle repair is expensive.
The immune system will not authorize it if:
- energy availability is erratic
- sleep is fragmented
- cortisol remains elevated
- circadian signals are inconsistent
This is why:
- under-eating blunts anabolic response
- sleep loss directly reduces protein synthesis
- chronic stress accelerates sarcopenia even with training
Repair happens only when the system perceives margin.
5. Time — the Most Ignored Variable
Immune systems remember. Chronic states do not reverse overnight.
In sarcopenia:
- macrophage tone shifts over weeks
- satellite cell responsiveness returns gradually
- ECM remodeling takes months
This is why early gains often feel neurological or energetic before they look structural.
The signal changes first, then the tissue follows.
Why This Reframes Treatment Entirely
Once sarcopenia is understood as an immune decision:
- training becomes a signal, not a punishment
- nutrition becomes support, not persuasion
- recovery becomes strategic, not optional
And most importantly: Failure to gain muscle is no longer regarded as personal failure. It is reframed as signal mismatch.
The Ethical Implication
Telling patients to “eat more protein and lift heavier” without addressing immune tone, recovery, and systemic stress is not just ineffective. It is unfair.
Muscle loss is not a lack of will but a lack of permission from the body to prioritize growth and maintenance.

Closing Takeaway
Sarcopenia and cachexia are not problems of muscle but problems of immune judgment.
Muscle is preserved when the immune system believes:
- the organism is safe
- the tissue is useful
- repair will pay off
Change those beliefs, and muscle follows.
PLAY
Someone asked me what I had spent my weekend doing. I told her that I’d worked on Wellth-e. What do you do for fun? she asked.
That is fun, I thought. Working on Wellth-e is like making scrapbooks on some of my favourite topics. When I first realized that I had OTS the exhaustion was so great that I slept in for two days and couldn’t really do anything for the rest of the week. But one day, I started reading again, and making notes for myself. And then I decided to put the notes together for an issue of Wellth-e.
I started listening to music again, to dance a little in my kitchen even though my hip still hurt. I pulled out books I hadn’t read in decades and made a decision that I was only going to do things I enjoyed–just for me.
When I’m working on Wellth-e, or listening to music, or watching the sunset, I get lost in the process I’m engaged in. I say Yes only to things that really matter to me. I stop a few times in the day just to lie down. When I do, I realize how tired my body is still, and I rest. I’m trying to learn to feel that fatigue when I’m not lying down, when I’m going about my day and in the middle of work. Because if I can feel it then, I can learn to stop. I can sit down for a bit and listen to my body’s aches and pains which are just requests for recovery time, healing time.
When I had my dog, Tula, I bought her toys and the both of us sat and stared at them. She was a puppy mill rescue and she looked to me for cues–what are we supposed to do here? I threw a ball and it was a chore for both of us to climb through some thick reeds to retrieve it. Neither of us felt like doing it again.
The two of us didn’t know how to ‘play’ but we sat in silence together a lot. She fell asleep in my arms when I was reading. We went for long walks by the river and went to dance class together. We enjoyed long rides to nowhere. I’d get on the Mass Pike and just drive for an hour or two before turning around and going home.
I know my idea of Fun and Play isn’t very appealing to most people and may have come across as oddball or strange. The projects I’ve tried and the experiments I’ve run have often puzzled people, and the curiosities in life that have awakened wonder in me have made some people shake their heads. But I’m just here right now with the sun streaming the most beautiful golden light into the room and Glenn Gould humming in the background as he plays Bach.
And life is so delightful.

References
Tidball, J. G. (2017). Regulation of muscle growth and regeneration by the immune system. Nature Reviews Immunology, 17(3), 165–178.
https://doi.org/10.1038/nri.2016.150
Howard, E. E., Pasiakos, S. M., Blesso, C. N., Fussell, M. A., & Rodriguez, N. R. (2020). Divergent roles of inflammation in skeletal muscle recovery from injury. Frontiers in Physiology, 11, 87.
https://doi.org/10.3389/fphys.2020.00087
Mosser, D. M., & Edwards, J. P. (2008). Exploring the full spectrum of macrophage activation. Nature Reviews Immunology, 8(12), 958–969.
https://doi.org/10.1038/nri2448
Murray, P. J., et al. (2014). Macrophage activation and polarization: Nomenclature and experimental guidelines. Immunity, 41(1), 14–20.
https://doi.org/10.1016/j.immuni.2014.06.008
Arnold, L., et al. (2007). Inflammatory monocytes recruited after skeletal muscle injury switch into anti-inflammatory macrophages to support myogenesis. Journal of Experimental Medicine, 204(5), 1057–1069.
https://doi.org/10.1084/jem.20070075
Yin, H., Price, F., & Rudnicki, M. A. (2013). Satellite cells and the muscle stem cell niche. Physiological Reviews, 93(1), 23–67.
https://doi.org/10.1152/physrev.00043.2011
Bentzinger, C. F., Wang, Y. X., & Rudnicki, M. A. (2013). Building muscle: Molecular regulation of myogenesis. Cold Spring Harbor Perspectives in Biology, 4(2), a008342.
Urciuolo, A., et al. (2013). Collagen VI regulates satellite cell self-renewal and muscle regeneration. Nature Cell Biology, 15(6), 637–646.
https://doi.org/10.1038/ncb2736
Gillies, A. R., & Lieber, R. L. (2011). Structure and function of the skeletal muscle extracellular matrix. Muscle & Nerve, 44(3), 318–331.
Kragstrup, T. W., et al. (2018). Fibrosis and altered ECM signaling in muscle aging. Frontiers in Aging Neuroscience, 10, 125.
von Roth, P., et al. (2014). Mesenchymal stem cell therapy for muscle regeneration: Role of the niche. Stem Cells International, 2014, 438140.
Gnecchi, M., et al. (2016). Paracrine mechanisms in adult stem cell signaling. Circulation Research, 118(6), 1084–1096.
https://doi.org/10.1161/CIRCRESAHA.116.303324
Bodine, S. C., et al. (2001). Identification of ubiquitin ligases required for skeletal muscle atrophy. Science, 294(5547), 1704–1708.
https://doi.org/10.1126/science.1065874
Sandri, M. (2008). Signaling in muscle atrophy and hypertrophy. Physiology, 23(3), 160–170.
Bonaldo, P., & Sandri, M. (2013). Cellular and molecular mechanisms of muscle atrophy. Disease Models & Mechanisms, 6(1), 25–39.
https://doi.org/10.1242/dmm.010389
Argilés, J. M., Busquets, S., Stemmler, B., & López-Soriano, F. J. (2014). Cachexia and sarcopenia: Mechanisms and potential targets. Current Opinion in Pharmacology, 22, 100–106.
Tisdale, M. J. (2009). Mechanisms of cancer cachexia. Physiological Reviews, 89(2), 381–410.
https://doi.org/10.1152/physrev.00016.2008
Thomou, T., et al. (2017). Adipose-derived circulating microRNAs regulate gene expression in other tissues. Nature, 542(7642), 450–455.
https://doi.org/10.1038/nature21365
Crewe, C., An, Y. A., & Scherer, P. E. (2017). The ominous triad of adipose tissue dysfunction. Journal of Clinical Investigation, 127(1), 74–82.
Sabaratnam, R., et al. (2020). Extracellular vesicles in muscle wasting and cachexia. Journal of Cachexia, Sarcopenia and Muscle, 11(3), 671–684.
Fearon, K., Strasser, F., Anker, S. D., et al. (2011). Definition and classification of cancer cachexia. The Lancet Oncology, 12(5), 489–495.
https://doi.org/10.1016/S1470-2045(10)70218-7
Baracos, V. E., Martin, L., Korc, M., Guttridge, D. C., & Fearon, K. C. H. (2018). Cancer-associated cachexia. Nature Reviews Disease Primers, 4, 17105.
https://doi.org/10.1038/nrdp.2017.105
Serhan, C. N., et al. (2015). Resolution of inflammation: State of the art. Nature Reviews Immunology, 15(8), 439–454.
https://doi.org/10.1038/nri3872
Tidball, J. G., & Villalta, S. A. (2010). Regulatory interactions between muscle and the immune system. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology, 298(5), R1173–R1187.
Need the bottom line on these new insights to muscle health? Read Postscript next and get the heads-up on your upcoming weekly action lists.
Explore more from Issue #3: New Insights into Muscle Health
Pick the next section to read in Issue 3
